New electrocatalyst reported in NPJ 2D Materials and Applications
17 01 2026
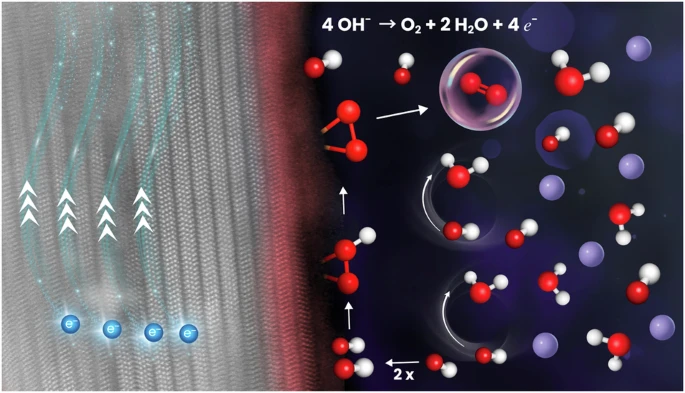
A team led by Hermenegildo Garcia at the Instituto de Tecnología Química, with contributions from our group at the University of Warsaw, reports a new MXene-based electrocatalyst that improves oxygen evolution performance. The NiFe–Ti₃C₂Clₓ nano-hybrid, obtained through a one-step molten salt synthesis, shows optimal activity at a 1:1 Ni:Fe ratio, reaching 310 mV overpotential and a low Tafel slope. The results demonstrate how controlled composition and nano-structuring of MXene-supported bimetallic phases enhance catalytic efficiency.
Density functional theory was essential in explaining the O–O bond formation pathway. The calculations compare the adsorbate evolution mechanism (AEM) and the lattice oxygen mechanism (LOM), showing that AEM dominates for both Ni and Fe active sites, with Ni being more active. The analysis links this preference to lower energy barriers for O–O coupling and favorable electronic structure at Ni sites, providing a consistent picture that connects experimental activity trends with atomistic reaction steps.
Kruger, D.D., Recio, F.J., Wlazło, M., Osella, S., Primo, A. & Garcia, H. 2026, “Facile molten salt synthesis of bimetallic NiFe-Ti3C2Tx MXene nano-hybrid as an efficient oxygen evolution electrocatalyst”, NPJ 2D Materials and Applications, vol. 10, no. 1, pp. 24.
